
Over 16,548,505 people are on fubar.
What are you waiting for?

Hydroxycut Recall after FDA Warning to Stop Using the Diet Pill
Following the death of a 19-year-old male and 23 reports of liver damage, the FDA has stepped in to warn consumers to discontinue use of the popular diet pill Hydroxycut. Hydroxycut manufacturer Iovate Health Sciences has agreed to recall 14 products from the market as “an abundance of caution.”
From the FDA press release — The FDA has received 23 reports of serious health problems ranging from jaundice and elevated liver enzymes, an indicator of potential liver injury, to liver damage requiring liver transplant. One death due to liver failure has been reported to the FDA. Other health problems reported include seizures; cardiovascular disorders; and rhabdomyolysis, a type of muscle damage that can lead to other serious health problems such as kidney failure.
The teen’s death happened in 2007, and was only reported to the FDA in March 2009. Dr. Linda Katz of the FDA’s food and nutrition division said it has taken so long to bring this to the public’s attention because the cases were rare and the FDA has no authority over supplements (a government report in March 2009 called for higher supervision of supplements by the FDA). “Part of the problem is that the FDA looks at dietary supplements from a post-market perspective, and an isolated incident is often difficult to follow,” she said.
The FDA does not regulate supplements with the same authority it does pharmaceuticals. Producers do not have to have an FDA approval nor inspection of the products for safety or efficacy before being sold to consumers. “You really have to be careful about dietary supplements, especially weight-loss pills,” said Ano Lobb, who has studied Hydroxycut for Consumer Reports. “People believe that the FDA has verified that these products are at least safe and effective, and that’s really not the case. When you see fantastic claims — that’s generally what they are.”
The FDA does monitor aftermarket reports for health problems related to these supplements. A similar warning in 2004 regarding ephedra forced the government to enact a ban on the supplement ingredient following reports of heart attack and stroke.
This list of Hydroxycut products being recalled include:
Hydroxycut Regular Rapid Release Caplets
Hydroxycut Caffeine-Free Rapid Release Caplets
Hydroxycut Hardcore Liquid Caplets
Hydroxycut Max Liquid Caplets
Hydroxycut Regular Drink Packets
Hydroxycut Caffeine-Free Drink Packets
Hydroxycut Hardcore Drink Packets (Ignition Stix)
Hydroxycut Max Drink Packets
Hydroxycut Liquid Shots
Hydroxycut Hardcore RTDs (Ready-to-Drink)
Hydroxycut Max Aqua Shed
Hydroxycut 24
Hydroxycut Carb Control
Hydroxycut Natural
Hydroxycut Cleanse and Hoodia products are not affected by the recall.
“The agency has not yet determined which ingredients, dosages, or other health-related factors may be associated with risks related to these Hydroxycut products. The products contain a variety of ingredients and herbal extracts,” per the FDA press release.
Chantix has become the leading prescription to help smokers kick the habit. Pfizer, Manufacturer of Chantix, has reported skyrocketing sales since its introduction in 2006. Sales totaled $101 million in 2006, $883 million in 2007, and $846 in 2008. It’s dominant in its market, with a reported 90% market share.
Chantix is unique because it not only reduces the urge to smoke, it actually blocks the brain’s nicotine receptors. This means if the smoker lapses and lights up, it won’t satisfy the nicotine craving. Chantix can be taken up to 12 weeks, by which time the smoker should have lost any urge to smoke. By design, Chantix users experience the similar symptoms of quitting smoking cold turkey. Many patients experience mood swings, depression, and even thoughts of suicide. Pfizer has stated these side effects are a result of nicotine withdrawal, and not the drug itself. Other’s believe there is a link to Chantix and the psychological side effects.
Since Chantix was approved just a little more than three years ago in May of 2006, considerable controversy has mounted. Within months of its approval, the FDA issued public warnings about the increasing likelihood of Chantix being associated with serious changes in patients' behavior including agitation, hostility, depression, suicidal thoughts, and reports of attempted and completed suicide. In many cases, the problems began shortly after starting the medication and ended when the mediation was stopped. In 2007, the FDA released an alert to doctors to look for unusual behavior. This came shortly after the musician Carter Albrect was shot and killed. Those close to him cited his use of Chantix and atypical behavior leading up to his death.
The FDA issued a Public Health Advisory on February 1, 2008, to alert patients, doctors, and caregivers to important new safety warnings on the drug label relating to suicidal behavior and several other neuropsychiatic symptoms. Also, in 2008, the FAA banned the use of Chantix by pilots and co-pilots, citing possible psychological side effects. Also in 2008, the Department of Veterans Affairs made headline news by issuing Chantix to soldiers with post-traumatic stress disorder. Earlier this year Health Canada (Canada’s Public Health Department) revised the warnings of Chantix after receiving over 800 reports of adverse psychological side effects. Most recently, the FDA has (finally) required Pfizer to place new warnings in the form of a Boxed Warning – the strongest warning possible – highlighting the risks concerning Chantix and a link to changes in behavior, hostility, agitation, depressed mood, suicidal thoughts and behavior, and attempted suicide. The FDA has instructed doctors to have their patients cease use immediately if they experience any of these side effects.
With hundreds of suicides reportedly linked to Chantix, and thousands more attempted suicides, Pfizer has gone far too long downplaying the psychological side effects as simply nicotine withdrawal.
There are other side afects thay don't want anyone to know about such as stomac bleeding and paine from long term use..
If you know of any other's please let me know so I may warn other's
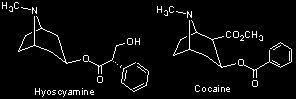 The action of tropane alkaloids at the cellular level is complex and is related to their molecular structure, particularly the methylated nitrogen at one end of the molecule (see tropane). This chemical structure is also found in the neurotransmitter acetylcholine, which transmits impulses between nerves in the brain and neuromuscular junctions. The anesthetic properties of tropane alkaloids may relate to their interference with acetylcholine, perhaps by competing with it at the synaptic junctions, thus blocking or inhibiting nerve impulses. It is interesting to note that the infamous tropane alkaloid cocaine, from the leaves of the coca shrub (Erythroxylum coca--Erythroxylaceae), is also a local anesthetic when injected into skin or muscle tissue. This property led to the discovery and synthesis of the more potent compound, novocain, widely used in dentistry
The action of tropane alkaloids at the cellular level is complex and is related to their molecular structure, particularly the methylated nitrogen at one end of the molecule (see tropane). This chemical structure is also found in the neurotransmitter acetylcholine, which transmits impulses between nerves in the brain and neuromuscular junctions. The anesthetic properties of tropane alkaloids may relate to their interference with acetylcholine, perhaps by competing with it at the synaptic junctions, thus blocking or inhibiting nerve impulses. It is interesting to note that the infamous tropane alkaloid cocaine, from the leaves of the coca shrub (Erythroxylum coca--Erythroxylaceae), is also a local anesthetic when injected into skin or muscle tissue. This property led to the discovery and synthesis of the more potent compound, novocain, widely used in dentistry
 Incas of the South American Andes have used coca leaves since pre-Columbian times for religious and medicinal purposes. They chew coca leaves for its stimulant properties to ward off fatique and hunger, to enhance endurance in the high altitude Andes, and to promote a sense of well-being. Cocaine acts primarily by interfering with the reuptake of dopamine. Dopamine produces feelings of well-being when it is released from one transmitting brain neuron to another. After release, dopamine is normally pumped back into the original neuron. Cocaine prevents the reuptake of dopamine and therefore prolongs the feeling of well-being.
Spanish conquistadores tried to prohibit coca use until they realized that the Indians they enslaved would work harder if allowed to chew the leaves. Coca leaves were taken back to Europe by the Spanish and in 1860 cocaine was extracted and became a popular drug in Europe. It was a popular ingredient in some beverages and medicines in the United States, including Coca Cola; however, since 1904 federal law has prohibited the inclusion of cocaine in any beverage. The Coca Cola Company complied with this law, but was sued for misleading advertising because the name implied that the beverage contained coca products. As a result, coca leaves, with the cocaine removed, were used to flavor the syrup from which the soda is made. In 1914 cocaine was formally declared illegal by the Harrison Narcotics Act.
Cocaine is a 3-ring alkaloid that is most commonly taken as a hydrochloride salt known as "coke." Crystals of cocaine hydrochloride are ground into a fine powder. One method of taking this drug is to sniff (snort) a fine line of the powder. Since the hydrochloride salt decomposes at the temperature required to vaporize it, a method was developed to convert cocaine into the liberated "base" form. "Free-base" cocaine was typically produced by heating the hydrochloride in the volatile solvent ether; however, since ether is very flammable, this method is hazardous. Crack cocaine is produced by heating the hydrochoride in a solution of baking soda until the water evaporates. This type of base-cocaine makes a cracking sound when heated; hence the name crack cocaine. Both freebase and crack cocaine can be injected and, since they are unaffected by heat, can be smoked.
During the past century, many famous people in Europe and the United States took cocaine regularly. The fictional Sherlock Holmes character quite openly took cocaine in several of the stories in order to stimulate and clarify the mind. Cocaine addiction is associated with a number of health risks. If inhaled for long periods of time, it can cause damage to the inner surface of the nose. Cocaine users tend to increase the dosage to maintain the original highs, and continual use may eventually cause damage to neuroreceptors.
Depending on the dosages, several tropane alkaloids of Datura and Brugmansia (when absorbed together) may have synergistic properties resulting in extreme hallucinations, delirium and death. Since the alkaloids are fat soluble they are readily absorbed through the skin and mucous membranes. Volumes have been written about the uses and properties of Datura in the Middle Ages. Most of the uses involved the consumption of potions or concoctions made from various parts of the plant. The famous seventeenth century Dutch artist, David Teniers the Younger, made several paintings of witches preparing for their demonic orgy or sabbat. The scenes frequently depicted a witch being anointed while she straddled a broom for her flight into the sky. Clay tablets from Babylonian and Assyrian ruins indicate that Datura was used medically in ancient civilizations several thousand years ago. Greek and Roman physicians used Datura mixed with opium as a sedative and general anesthetic during surgery. In fact, the use of scopolamine (one of the alkaloids in Datura) plus morphine as an effective pain reliever and sleep inducer was common practice until the nineteenth century.
Incas of the South American Andes have used coca leaves since pre-Columbian times for religious and medicinal purposes. They chew coca leaves for its stimulant properties to ward off fatique and hunger, to enhance endurance in the high altitude Andes, and to promote a sense of well-being. Cocaine acts primarily by interfering with the reuptake of dopamine. Dopamine produces feelings of well-being when it is released from one transmitting brain neuron to another. After release, dopamine is normally pumped back into the original neuron. Cocaine prevents the reuptake of dopamine and therefore prolongs the feeling of well-being.
Spanish conquistadores tried to prohibit coca use until they realized that the Indians they enslaved would work harder if allowed to chew the leaves. Coca leaves were taken back to Europe by the Spanish and in 1860 cocaine was extracted and became a popular drug in Europe. It was a popular ingredient in some beverages and medicines in the United States, including Coca Cola; however, since 1904 federal law has prohibited the inclusion of cocaine in any beverage. The Coca Cola Company complied with this law, but was sued for misleading advertising because the name implied that the beverage contained coca products. As a result, coca leaves, with the cocaine removed, were used to flavor the syrup from which the soda is made. In 1914 cocaine was formally declared illegal by the Harrison Narcotics Act.
Cocaine is a 3-ring alkaloid that is most commonly taken as a hydrochloride salt known as "coke." Crystals of cocaine hydrochloride are ground into a fine powder. One method of taking this drug is to sniff (snort) a fine line of the powder. Since the hydrochloride salt decomposes at the temperature required to vaporize it, a method was developed to convert cocaine into the liberated "base" form. "Free-base" cocaine was typically produced by heating the hydrochloride in the volatile solvent ether; however, since ether is very flammable, this method is hazardous. Crack cocaine is produced by heating the hydrochoride in a solution of baking soda until the water evaporates. This type of base-cocaine makes a cracking sound when heated; hence the name crack cocaine. Both freebase and crack cocaine can be injected and, since they are unaffected by heat, can be smoked.
During the past century, many famous people in Europe and the United States took cocaine regularly. The fictional Sherlock Holmes character quite openly took cocaine in several of the stories in order to stimulate and clarify the mind. Cocaine addiction is associated with a number of health risks. If inhaled for long periods of time, it can cause damage to the inner surface of the nose. Cocaine users tend to increase the dosage to maintain the original highs, and continual use may eventually cause damage to neuroreceptors.
Depending on the dosages, several tropane alkaloids of Datura and Brugmansia (when absorbed together) may have synergistic properties resulting in extreme hallucinations, delirium and death. Since the alkaloids are fat soluble they are readily absorbed through the skin and mucous membranes. Volumes have been written about the uses and properties of Datura in the Middle Ages. Most of the uses involved the consumption of potions or concoctions made from various parts of the plant. The famous seventeenth century Dutch artist, David Teniers the Younger, made several paintings of witches preparing for their demonic orgy or sabbat. The scenes frequently depicted a witch being anointed while she straddled a broom for her flight into the sky. Clay tablets from Babylonian and Assyrian ruins indicate that Datura was used medically in ancient civilizations several thousand years ago. Greek and Roman physicians used Datura mixed with opium as a sedative and general anesthetic during surgery. In fact, the use of scopolamine (one of the alkaloids in Datura) plus morphine as an effective pain reliever and sleep inducer was common practice until the nineteenth century.